Autologous biologics live or die on documentation. PPX™ standardizes the manufacturing and documents every prep. Every time.
i · Autologous
Patient's own.
PPX™ starts with the patient's own platelets — drawn chair-side. Fully autologous means no donor concerns, no allogenic regulatory questions, no compatibility issues.
ii · Concentrated
Just the EVs.
Multi-step centrifugation removes red cells, leukocytes, and plasma debris. What's left is the concentrated platelet-derived EV fraction — the cargo that carries growth factors and microRNA.
iii · Documented
COA per prep.
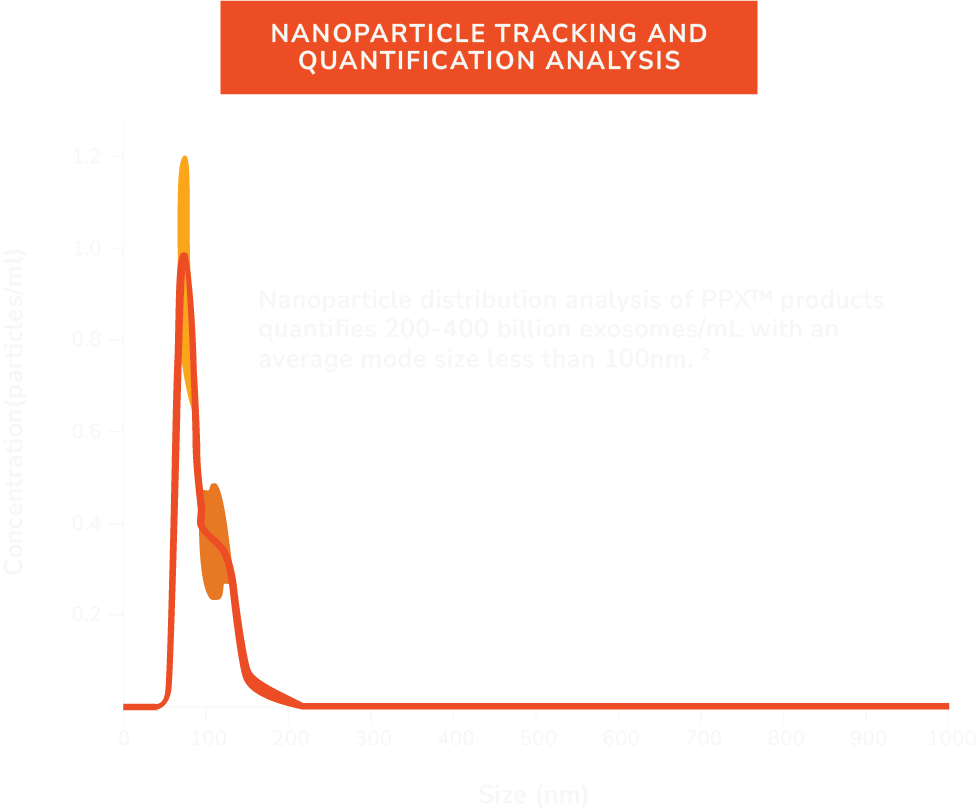
Every preparation ships with a certificate of analysis documenting sterility, identity, and nanoparticle count — confirming exactly what's in the vial before it reaches the patient.
iv · Chair-Side
Office-ready.
A standard chair-side blood draw is sealed and shipped overnight to an FDA-registered, cGMP-compliant lab. Multi-step centrifugation isolates the EV fraction. Final product ships back in two 1mL vials, ready to inject.